Adeno-associated viruses have taken the gene therapy field to new heights, largely because of it’s reduced immunogenicity compared to its adenoviral cousin. But while the field has made tremendous strides in creating better, safer viruses, it still remains imperative that batches be held to the highest quality standards if they’re to be given to human patients. Part of that process is understanding the viral titer through the production process to be able to accurately dose patients. Dose too little and you might not see a response, dose too much and you could run into potentially dangerous side effects.
A traditional way that scientists have quantified titer has been the tried and true Real Time PCR (qPCR) method. While this process certainly works, especially in an academic research lab setting, the need for a standard curve means that you can only semi-quantify, and your data are only as good as your curve.
New technology has paved the way to analyze AAVs much more accurately, with QIAGEN leading the charge in the space. QIAGEN has developed a CGT Viral Vector Lysis Kit that, when paired with their QIAcuity® Digital PCR System results in both a standardized prep workflow and absolute and precise quantification of viral titers, without the need for a standard curve. The kit is suitable for the lysis of AAV2, AAV5, AAV6, AAV8 and AAV9 and also has multiplexing capabilities.
We know there’s always a bit of hesitancy when delving into a new kit and technology – that’s why the QIAGEN team, in partnership with North Dakota-based Agathos Biologics, is presenting data at this week’s ASCGT conference.
“Measuring viral vectors is notoriously challenging, especially using qPCR,” said James Brown, PhD, CEO and Co-Founder of Agathos. “We purchased the QIAcuity dPCR instrument in late 2021 after looking at several competitors, and we worked closely with QIAGEN to come up with better solutions to measure AAV titers for our work.”
“It was a really synergistic relationship – our team and their team shared data back and forth easily. It was everything you think a partnership ought to be but rarely is,” Brown added. “We hope that Agathos and QIAGEN can continue to grow together so that we can get closer to the ultimate goal – to deliver impactful gene therapies for patients.”
How does it work?
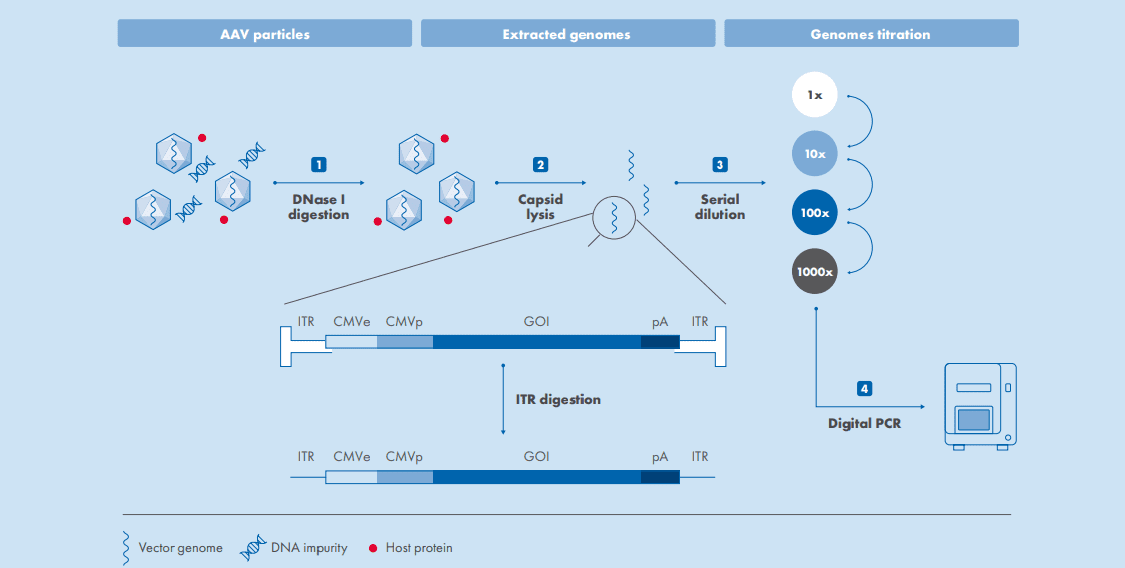
The workflow uses four standardized steps to seamlessly prep the sample for dPCR:
- Cell lysate samples are treated with DNaseI to remove any DNA impurities
- Viral capsids are lysed with or without Proteinase K, revealing the vector genome
- Lysates are serially diluted
- Samples are loaded onto the QIAcuity dPCR instrument to determine titer

Do different types of cell lysates affect the process and results?
In the data to be presented at ASCGT, the teams tested several types of cell lysates from cells commonly used for AAV production to see if the type of cell affected viral vector titer quantification. The clarified cell lysates for Sf9, Tnms42, HEK293, and BHK-21 did not significantly impact quantification of an AAV2 reference standard.
Results are reliable and reproducible
Also in the presentation, the team quantified the genome titer of AAV5 particles over a series of three days between different operators and laboratories and found there was little variation, further validating the consistency of the process and results.
Interested in seeing the data for yourself? Check out more info on their poster below:
- “Novel CGT Viral Vector Lysis Kit for AAV Enables a Standardized AAV Workflow from Cell Lysate to Absolute and Precise Quantification of Viral Titers”, Abstract Number 1491.
Agathos also presented two posters earlier this week – you can find more info below: