A Personal Connection Fuels Commitment to Advancing Cystic Fibrosis Treatment for ISPE Keynote Speaker
Many pharma execs are proud to be part of an industry that advances life-saving treatments, giving hope and easing suffering for millions of families and individuals. For Alan Holmer, Director of Corbus Pharmaceutical Holdings, Inc, and Chairman of the Board at the Metropolitan Washington D.C. Chapter of the Cystic Fibrosis Foundation (CFF), this pride and commitment to saving lives goes double: both of his children are alive today because of advances in Cystic Fibrosis (CF) treatment.
Holmer’s keynote address at the recent ISPE Chesapeake Bay Chapter’s Mid-Atlantic Science and Technology (MAST) showcase provided unique insight into one of the great medical stories of our lifetime: how, through venture philanthropy, the CFF has been instrumental in giving millions of individuals with CF a new lease on life.
Holmer began his speech by taking the audience back to the spring of 1980 when he and his wife received life-altering news: their infant son was diagnosed with CF, with a life expectancy of 18. Despite this tragic diagnosis, he and his wife rolled the dice three years later and had another child; she, too, had CF and shared a similarly-short lifespan.
When the CFF was founded in 1955, life expectancy for those with the disease was less than five years. Ever since, the CFF has remained dedicated to raising funds to fuel CF research and drug development programs in partnership with pharmaceutical companies. Sixty-three years later, people with CF are living into their 30s, 40s and beyond as a direct result of CFF’s support.
The CFF is also in a better position today to help raise that life expectancy even more and eventurally cure CF for all patients with nearly $4B in the bank. CFF became one of the wealthiest medical foundations as a result of a strategic decision on how they applied their money to help develop cures for CF through a venture philanthropy model. After investing $150M over the year into a young drug development company with very promising technology, Vertex Pharmaceuticals, Inc., they also took an equity position in the company in return. When Vertex received FDA approval in 2012 CFF began seeing annual royalty payments in the $200-$500M rage before selling those royalties to a NY firm in 2014 for a $3.3B payout.
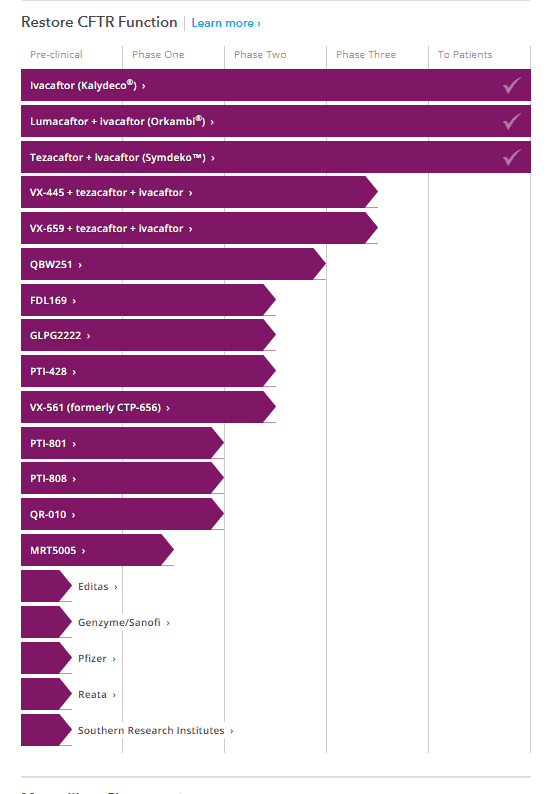
There are now three (3) FDA approved CF drugs that restore CFTR function for CF patients and nearly a dozen more in clinical trials. A dozen other approved drugs now address anti-inflamatory, Mucucilary, Anti-infective and GI conditions that improve the health and life of patients. CF is a genetic disease though so each of these drugs only addresses a segment of the CF population. This is where Holmer emphasized the opportunity and need for more funding and research to address. The emergence of gene and cell-based therapies and personalized medicine approaches to CF are expected to have a promising impact the still unaddressed CF populations.
See full drug development pipeline at https://www.cff.org/trials/pipeline
Holmer shared how both his children benefited through the years from new therapies as they came to market, and how both are now living productive lives: his son a Catholic priest and his daughter a licensed clinical social worker. He shared a quote from a speech his daughter, Joy, recently made, in which she talked about what it was like to live with CF:
“When my older brother was diagnosed with CF in 1980, my mom prayed the rosary every day for a year, asking God to spare me from having CF. God answered her prayer: his answer was no. I personally think God’s pretty smart, because having CF has been a blessing. It’s allowed me to develop an incredible relationship with my CF partner-in-crime, my older brother. It has taught me resilience, empathy, compassion and, most importantly, to never take life for granted.”
Holmer concluded by emphasizing that, despite the advances in treating CF, the fight is far from over. CFF will continue fundraising and investing in research and drug development until they achieve their ultimate goal: a cure for all people with CF.
[Visit CFF’s Drug Development Pipeline to learn more about the CF drugs in development or already in use due to CFF’s support]
Author:
- About the Author
- Latest Posts
Over the past 11 years, Chris has grown BioBuzz into a respected brand that is recognized for its community building, networking events and news stories about the local biotech industry. In addition, he runs a Recruiting and Marketing Agency that helps companies attract top talent through a blended model that combines employer branding and marketing services together with a high powered recruiting solution.