The battle against COVID-19 and to flatten the curve is ongoing across the globe. While there have been some signs of hope coming out of Wuhan, China and New York recently, a long, tough fight remains as cities, towns and communities progress through their own respective COVID-19 curves.
Johns Hopkins was among the first institutions to respond to this crisis, having had considerable experience and resources dedicated to infectious disease preparedness. The Johns Hopkins Hospital Biocontainment Unit is one of 10 regional centers across the country designed to respond to outbreaks of highly infectious diseases.
From its Global COVID-19 dashboard and telehealth tools to coronavirus diagnostic test development and 3-D printed ventilator designs, Johns Hopkins remains at the forefront of the fight against COVID-19 with a number of projects in process across a wide range of coronavirus pandemic response areas. The institutional innovation and responsiveness highlights Johns Hopkins leadership role at the forefront of biomedical research and healthcare.
Here’s a roundup of several innovative COVID-19 programs that Johns Hopkins has launched since the crisis began:
Global COVID-19 Tracker
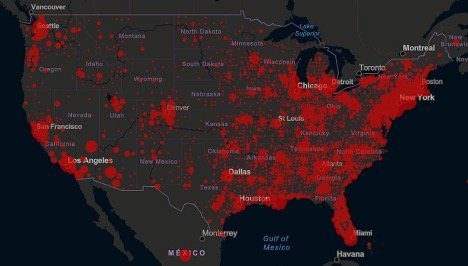
From the very early days of the crisis, a small team from the Johns Hopkins Center for Systems Science and Engineering stepped out front to track COVID-19’s spread across the globe. The dashboard, which has become the foremost source of up to date used daily by millions around the world, was born from a casual conversation between Lauren Gardner, associate professor of civil and systems engineering at Johns Hopkins, and one of her first-year Ph.D. advisees, Ensheng Dong.
Within 24 hrs Dong had created a live dashboard that aggregates data from the World Health Organization (WHO) and the CDC (Center for Disease Control) to track the coronavirus pandemic in real time. The dashboard was one of the first to takes COVID-19 data and parses it out by the number of confirmed cases and deaths by country, region and by state within the U.S. The data was also made open and freely downloadable since the beginning.
The data is displayed on a map that allows the user to dig deeper into COVID-19 trends, including the number of active cases, the incident rate, the case-fatality ratio and testing and hospitalization rate. The data and many charts were made openly downloadable since the beginning.

The John Hopkins coronavirus dashboard has become an invaluable tool for the media, researchers and individuals to stay up-to-date on the latest COVID-19 data in an easy, reliable and user-friendly manner, and one of the most cited sources for information.
Innovating How They Monitor Healthcare Workers
Johns Hopkins has deployed a mobile health software tool developed by Baltimore, Maryland’s Emocha, an emerging telehealth company. Emocha’s telehealth tool is helping JHU monitor its healthcare workers for COVID-19 for 14 days after potential coronavirus exposure. The tool asks users to fill out a form describing their symptoms, including their temperature; check ups are conducted via asynchronous video sessions.
Emocha works with healthcare facilities to get lists of doctors, nurses and other staff that might have been exposed to COVID-19. The company then communicates via secure chat and even helps to triage more serious cases for referral to the appropriate health services.
The most effective method for protecting frontline healthcare workers would be universal testing and quarantining asymptomatic doctors and nurses. These approaches are not practical, however, as healthcare facilities don’t have the testing capacity and removing doctors and nurses from the rotation would mean fewer staff left to treat sick patients. Emocha’s telehealth tool helps Johns Hopkins doctors and nurses keep working while ensuring their health and wellbeing are being monitored closely and acted on immediately should more symptoms arise.
Johns Hopkins’ COVID-19 Diagnostic Test
In early March, Johns Hopkins doctors realized that national test shortages would prevent them from testing all of their patients in need, so they took it into their own hands to develop their COVID-19 diagnostic test.
The test, developed by Johns Hopkins’ clinical microbiologists Karen Carroll, M.D., and Heba Mostafa, M.B.B.Ch., Ph.D., was first used on March 11. Within three days, approximately 85 tests were performed. The test analyzes a nasal or oral swab and takes 24 hours to yield results, but Mostafa hopes she can shorten that to as little as three hours while ramping capacity up to as many as 1,000 tests per day.
Serological Test in Pre-clinical Development
Serology testing for SARS-CoV-2 is in high demand to better quantify cases of COVID-19, including identifying individuals that may be asymptomatic or have already recovered from the virus.
Lisa Maragakis, Senior Director of Infection Prevention with Johns Hopkins Medicine, citing data from Johns Hopkins Medicine, stated that 80 to 85 percent of people who are infected will have mild to no symptoms.
In contrast to the widely used RT-PCR COVID-19 tests that can only identify viral material only for individuals currently infected, Serology tests analyze a person’s immune response to diagnose if a person was infected and has subsequently recovered.
By identifying individuals with SARS-Cov- 2 antibodies, these tests could potentially be a path to identify Americans with these antibodies who can then opt out of social distancing restrictions and get back to work, which will help restart the economy.
The Klein lab at Johns Hopkins Bloomberg School of Public Health (JHSPH) is in pre-clinical development stages of a new enzyme-linked immunosorbent assay or ELISA serological test for SARS-Cov-2 antibodies. A team led by Dr. Fatima Amanat adapted an ELISA that tests for IgG and IgM to the full length Spike protein and to the receptor binding domain (RBD). They are now working to get a mucosal IgA ELISA working. So far, they are using the kit to test samples from Johns Hopkins Hospital.
Just this week NIH announced it was launching a 1,000 person serological survey, testing anti-SARS-CoV-2 on the US population. In fact, this study is the first serology study undertaken by NIAID.
COVID-19 Plasma-based therapy gets FDA Clinical Approval
On Friday April 3, a research collaboration led by Johns Hopkins University (JHU) received FDA approval for clinical testing of a plasma-based COVID-19 therapy to treat critically-ill patients, and as a means of preventing healthy people, especially first responders and healthcare workers, from getting sick. The plasma therapy uses plasma collected from COVID-19 patients that have recovered from the virus.
The FDA first started allowing researchers to request emergency authorization for its use on March 24th and within just a few days New York and Houston were starting treatments using the therapy. Since then the FDA has approved an expanded access program which will allow numerous other U.S. hospitals to follow suit, according to Aaron Tobian, the director of the Division of Transfusion Medicine at Johns Hopkins Hospital.
A second FDA approval is pending for a Hopkins clinical trial on patients who are slightly or moderately ill. The goal for this trial would be to keep patients out of ICUs and help them fully recover.
The research team from across the U.S. was assembled by JHU infectious disease expert Arturo Casadevall. The team consists of representatives from over 20 hospitals and research organizations, including the Albert Einstein College of Medicine, Stanford University Medical Center and the Mayo Clinic. The Red Cross and the New York Blood Bank will partner with the research group to collect plasma from people identified to have SARS-CoV-2
In late March, the research group received $4 million to fund this COVID-19 research, $3M of which was from Bloomberg Philanthropies and the remaining $1M from the State of Maryland.
Johns Hopkins Engineers Develop 3-D Printed Ventilator Splitters
A team of Johns Hopkins engineers has developed a 3-D printed ventilator splitter that could potentially solve safety and efficacy concerns around the splitting of ventilators.
Sung Hoon Kang, an assistant professor of mechanical engineering at the Johns Hopkins Whiting School of Engineering, is leading the team that also includes pulmonary and ICU specialists from the Johns Hopkins School of Medicine. COVID-19 patients suffering from acute respiratory distress syndrome (ARDS) require a ventilator to manage the build up of fluid in the lungs and the subsequent deprivation of the oxygen needed for organ function. Being able to more safely and effectively deploy split ventilators could help alleviate the shortage of ventilators and save more lives.
The new 3-D printed ventilator is designed to prevent two major split ventilator concerns: Cross contamination and a failure of the split ventilator to deliver the needed oxygen to both patients. The new design adds a filter to mitigate cross contamination and an air flow monitoring system to assist caregivers in the management of proper air flow to each patient.
Kang’s team hopes to start testing the new design in model lungs at some point after mid-April 2020. Once the design receives FDA approval, the team plans to release the design for public use.
Needless to say, the various teams at JHU are addressing this world-wide pandemic from multiple angles. From testing to vaccines to treatments, their leadership has become a beacon of hope that together with other institutions, organizations and government agencies, we will get through this together.