“5 Questions With…” is a weekly BioBuzz series where we reach out to interesting people in the BioHealth Capital Region to share a little about themselves, their work, and maybe something completely unrelated. This edition features 5 Questions with Dr. Amritha Jaishankar, Director, Programs and Strategic Alliances, Maryland Stem Cell Research Fund (MSCRF).
Dr. Amritha Jaishankar serves as the Director, Programs and Strategic Alliances, Maryland Stem Cell Research Fund (MSCRF). In this role, Amritha is responsible for oversight of programs, scientific management, and oversight of the portfolio, as well as MSCRF business development. She works with stem cell scientists across Maryland to develop a collaborative research program and promote the commercialization of stem cell technologies. MSCRF has invested over $165 million in identifying, supporting, and accelerating cutting-edge research, innovation, clinical trials, and commercialization of human stem cell-based technologies in Maryland.
Amritha brings over a decade of experience in stem cell research in federal, university, and industry settings. She is passionate about using stem cell technologies to accelerate cures for patients in need. She was recently recognized for her leadership in life sciences and contributions to the community as a 2020 Leading Women honoree by The Daily Record, and as Women In Bio Capital Region’s Rising Star.
1. Please introduce yourself to our audience with a look back on your career.
Thank you for the opportunity to do this. I am grateful to be a part of this vibrant life science community in the Maryland region for over ten years now and it has been great to see how this industry has evolved, and even more rewarding to have the ability to shape some of this growth in regenerative medicine. I currently serve as the Director of Programs and Strategic Alliances for the Maryland Stem Cell Research Fund (MSCRF) but I’ve been dedicated to advancing science my entire life.
I grew up in India where I specialized in biology since 10th grade, and I became very interested in understanding genetic diseases and gene therapy. I moved to the UK to gain hands-on research skills in this field, and upon graduating at the top of my Masters class there, I was offered a fellowship to pursue a Ph.D. in Genetics, which brought me to America.
I first started working with stem cells here and I was especially drawn to their role in brain development. This led me to the lab of one of the pioneers of cellular neurobiology at NIH where I started my postdoctoral work, and this gave me the opportunity to be a founding member of a non-profit institute at the Johns Hopkins School of Medicine where I worked in collaboration with AstraZeneca to advance treatments for neurodevelopmental disorders.
I have always been passionate about advancing therapies to patients in need, and I found myself at the intersection of science and business but still drawn to patient-oriented and mission-focused work.
I heard (like all good things… over a beer at a BioBuzz event!) that the Maryland Stem Cell Research Commission was looking for someone to spearhead an “Accelerating Cures” initiative and it took about 30 seconds for both of us to know it was a great fit.
A little over 4 years in, I have been fortunate to be able to make an impact and help accelerate cures in a field I am passionate about.
2. You are currently the Director, Programs and Strategic Alliances, Maryland Stem Cell Research Fund (MSCRF). Tell us about your current role and the program.
At MSCRF, we are focused on identifying and fostering cutting-edge research and innovation in the field of regenerative medicine in Maryland. Our mission is to develop new medical strategies for the prevention, diagnosis, treatment, and cure of human diseases, injuries, and conditions through human stem cells. We have six different programs to achieve this mission.
My role involves oversight and management of these programs and our portfolio, but what that entails honestly depends on the day and who I am interacting with! We have a diverse array of stakeholders including but not limited to our scientists, companies, physicians, and patients who drive what we do, our partners in this endeavor – the universities & research institutes, other stem cell agencies and foundations, investors, various players in the global cell therapy industry, local ecosystem collaborators like incubators and biohealth agencies and of course the legislators who support our efforts.
In addition, within each category we have further classifications, for example, we support early through clinical-stage companies, trainees, new and established researchers, and clinicians, etc. I have an active, hands-on award management model that allows us to build a strong relationship with each member of our portfolio so I can address each of their unique needs and enable them to succeed.
I am dedicated to identifying the most promising technologies and supporting each of them with the guidance and resources they need to get to the next stage and to take us one step closer to delivering a cure to a patient in need. We leverage our scientific and industry knowledge and alliances to create value and help each project we fund get to the finish line, and help our companies grow.
With almost 500 projects funded to date and having worked with leading scientists and key stakeholders in this industry, we have built a collaborative stem cell program and a strong stem cell community that I am so proud of, and we Accelerate Cures together.
3. What do you feel are the biggest goals for the MSCRF in 2021?
Our goal at MSCRF is to accelerate stem cell research, commercialization, and cures. Despite the pandemic, 2020 was a year of growth and record financing for regenerative medicine/advanced therapies. Our goal is to keep up this momentum and urgency to advance science and deliver cures.
In 2020, we funded 27 projects addressing a wide range of disease indications including COVID-19, Diabetes, Stroke, Heart disease, Cancer, Alzheimer’s disease, Parkinson’s disease, and more. Several of these conditions are amongst the top 10 leading causes of death in Maryland. We remain committed to translating scientific excellence and developing cures for these debilitating diseases.
We are proactive about identifying the next promising technology and company, and my priority is always to enable their success. As anyone in this field knows, every step is specialized and nuanced whether it is the science, manufacturing, delivery, regulatory, or pricing/reimbursement- we are innovating at each step.
I may be biased, but there is no other field poised to revolutionize healthcare the way cell and gene therapy is because we deliver cures, not just treatments. This requires a lot of resources and so we are looking to collaborate with those who want to help us advance these exciting, new technologies- whether it is to co-fund, follow-on fund, or provide us with complementary resources to accelerate cures. Please reach out to me to learn more.
2020 created unprecedented access and scientific collaboration and we will aim to maintain that level of engagement with our industry and key stakeholders.
We also have various internal operational and process/policy development goals because I aim to innovate, improve and deliver excellence, just as I expect from those we work with. Above all, we will continue to build this community and bring us all together with a sense of urgency and a common purpose to advance science and improve human health.
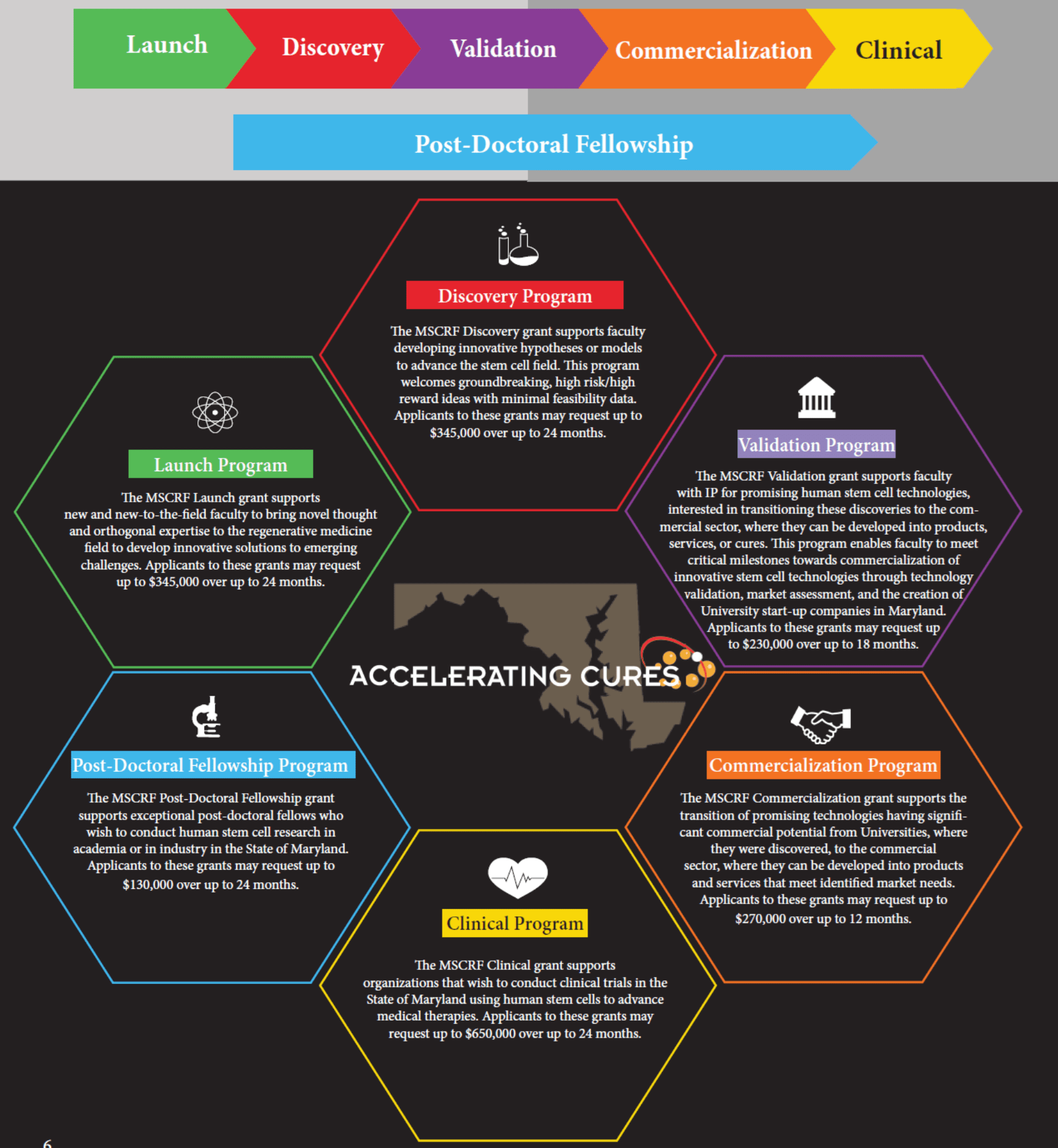
4. You offer a few different funding opportunities. Can you tell us a little about each one?
MSCRF offers 6 different funding opportunities- Launch, Discovery, Validation, Commercialization, Clinical, and Postdoctoral Fellowship.
Our programs are designed around what it takes to transition the most promising discoveries from the labs where the invention occurred to the clinic where they will be offered to patients, as well as a mechanism to train the next generation of stem cell scientists and leaders.
You can learn about each of our 6 programs and find a detailed RFA for each here or on the MSCRF App. I also encourage anyone interested in these programs to reach out to me to discuss their ideas and evaluate a fit.
Briefly, our commercialization program supports Maryland-based start-up companies or established companies developing a new stem cell product. Our Clinical program supports organizations that wish to conduct stem cell/cell therapy clinical trials in the State of Maryland. The Discovery and Validation programs support faculty advancing cutting-edge stem cell research and technologies. The Fellowship program supports stem cell research postdoctoral training for recent graduates. Last year we also created the Launch program to support new or new-to-the field faculty to bring novel thought and orthogonal expertise to the stem cell field. The MSCRF team is actively engaged with our stem cell community and we work with the Stem Cell Research Commission (our Board) to routinely evaluate and redesign our programs to address critical gaps and needs in the regenerative medicine industry.

5. What’s the most interesting thing about you that we wouldn’t learn from your resume or LinkedIn Profile?
Wait… and we post this on LinkedIn? 😉
Well, normally I’d be inclined to talk about one of my many interests, but we’ve all had a different year and are facing several more months of this, so while things I’ve done have looked different over the past year, the why hasn’t changed- I love connecting with people and learning and growing each day.
I don’t think anyone who has crossed paths with me will doubt my passion for my work or the people in my life, and this extends to most things I pursue. My friends would say my innate curiosity, authenticity and resilience keep it interesting- we always have a good time, can discuss almost any subject and I’m always ready for that next adventure.
Thank you to Dr. Amritha Jaishankar for participating in the ‘5 Questions with BioBuzz’ series and stay tuned for more interviews with others from across the BioHealth Capital Region and beyond.